What are protein domains?
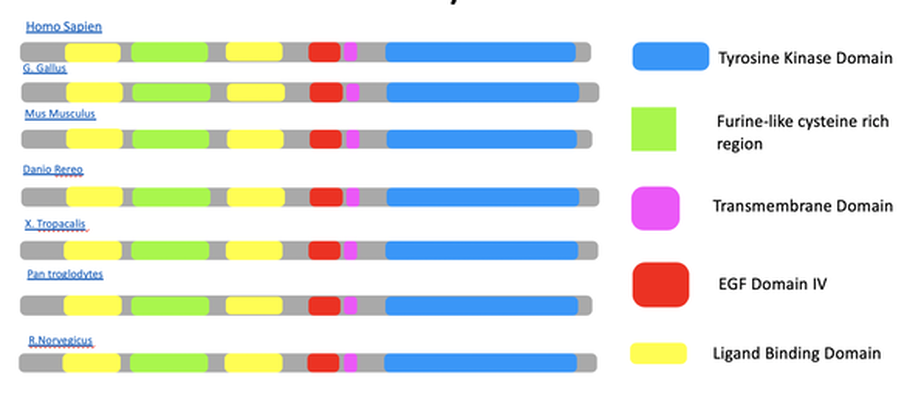
Protein domains fold independently and correspond to a specific function of the polypeptide. In EGFR, there are several domains that work in concert to maintain its function as a cell membrane receptor. To produce combinatorial mechanisms, protein domains function independently, which determines the protein’s overall function.
Protein motifs, on the other hand, are classified as super-secondary structures which are complexes of secondary structures [1]. These aren’t independent structures, only structural patterns that may occur at ligand or transmembrane domains. How do EGFR mutations correspond to peptide structure? Where do driver mutations reside and what implications does this have on function?
Protein motifs, on the other hand, are classified as super-secondary structures which are complexes of secondary structures [1]. These aren’t independent structures, only structural patterns that may occur at ligand or transmembrane domains. How do EGFR mutations correspond to peptide structure? Where do driver mutations reside and what implications does this have on function?
|
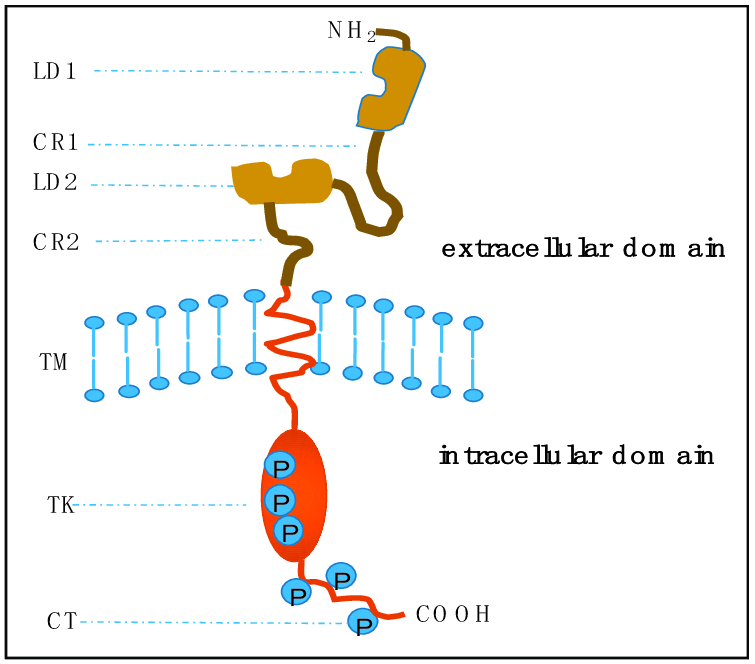
In the illustration, EGFR is shown in the plasma membrane in its monomer form. The three shares that occur on the peptide represent important protein domains including the tyrosine kinase, extracellular intracellular and domain. When EGFR dimerizes and undergoes its catalytic activity, the intracellular domain interacts with cytoplasmic molecules [1]. This causes signal cascades that effect cellular processes. In other words, understanding the protein domains of EGFR can yield important information about cell function.
|
Results
The epidermal growth factor receptor, also known as EGFR or HER1/ErbB1, is a member of the human EGFR tyrosine kinase family [2]. EGFR plays an essential role in signaling role in cell growth and is often hyper-activated in human cancers, which is especially important in vital organs, like the lung. This genes malignancy has made EGFR an important point of interest for researchers [3].
Fully active EGFR occurs in cells as a heterodimer. An EGFR monomer comprises of an N-terminal ligand-binding extracellular domain and an intracellular module connected by a single-pass transmembrane helix. The extracellular segment is made up of four domains (I–IV) which connects a small transmembrane segment and a tyrosine kinase domain (TKD). Ligand binding stabilizes the dimer conformation thereby causing an allosteric interaction, allowing Grb2 binding to the intercellular domain [4].
Discussion
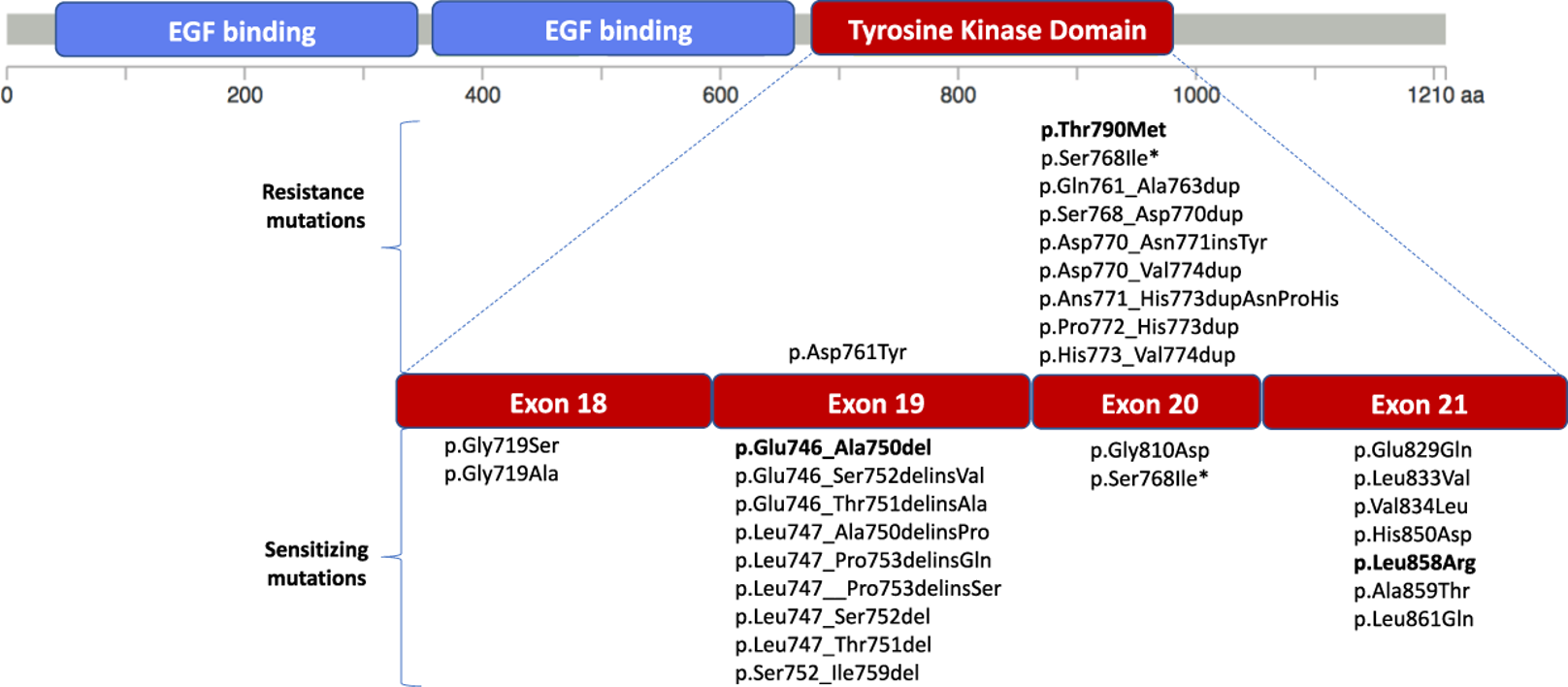
TK and TM domain mutations of EGFR seem to have profound mutations, some of which occur in LADC. However, mutations in the extracellular domains of EGFR may be important for understanding how TKIs can inhibit tumor cell growth. Tumor cells may express mutant forms of EGFRs protruding portion that could aid in prognosis as well as treatment if LADC. For example, A289V and R108K mutations in EGFR are related to oncogene behavior [4]. Although not necessarily studied against LADC, they may prove to be related to cell communication through EGFR.
References
1.) Zanetti-Domingues, Korovesis, et al, 2018, The architecture of EGFR’s basal complexes reveals autoinhibition mechanisms in dimers and oligomers. Retrieved From: https://www.nature.com/articles/s41467-018-06632-0
2.) Fernandez, Clarke, et al., Structure and Dynamics of the EGF Receptor as Revealed by Experiments and Simulations and Its Relevance to Non-Small Cell Lung Cancer (2019).
Retrieved from: https://europepmc.org/article/med/30959819
3.) Pirker, Hirsch, et al., Consensus for EGFR Mutation Testing in Non-small Cell Lung Cancer: Results from a European Workshop, 2010. Retrieved from: https://www.sciencedirect.com/science/article/pii/S1556086415318220
4.) Lee, Feng, et al.,Epidermal Growth Factor Receptor Activation in Glioblastoma through Novel Missense Mutations in the Extracellular Domain, 2016. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1702556/
2.) Fernandez, Clarke, et al., Structure and Dynamics of the EGF Receptor as Revealed by Experiments and Simulations and Its Relevance to Non-Small Cell Lung Cancer (2019).
Retrieved from: https://europepmc.org/article/med/30959819
3.) Pirker, Hirsch, et al., Consensus for EGFR Mutation Testing in Non-small Cell Lung Cancer: Results from a European Workshop, 2010. Retrieved from: https://www.sciencedirect.com/science/article/pii/S1556086415318220
4.) Lee, Feng, et al.,Epidermal Growth Factor Receptor Activation in Glioblastoma through Novel Missense Mutations in the Extracellular Domain, 2016. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1702556/